Journal Description
Vaccines
Vaccines
is an international, peer-reviewed, open access journal published monthly online by MDPI. The American Society for Virology (ASV) is affiliated with Vaccines and their members receive a discount on the article processing charges.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q1 (Immunology) / CiteScore - Q1 (Pharmacology (medical))
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 19.2 days after submission; acceptance to publication is undertaken in 2.9 days (median values for papers published in this journal in the second half of 2023).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
7.8 (2022);
5-Year Impact Factor:
7.4 (2022)
Latest Articles
Turkish Adaptation, Reliability, and Validity Study of the Vaccine Acceptance Instrument
Vaccines 2024, 12(5), 480; https://doi.org/10.3390/vaccines12050480 (registering DOI) - 29 Apr 2024
Abstract
This research study aimed to assess the reliability and validity of the Turkish version of the Vaccine Acceptance Instrument (VAI). The VAI is a 20-item Likert-type scale, with responses ranging across seven points. A systematic approach was followed to translate the scale into
[...] Read more.
This research study aimed to assess the reliability and validity of the Turkish version of the Vaccine Acceptance Instrument (VAI). The VAI is a 20-item Likert-type scale, with responses ranging across seven points. A systematic approach was followed to translate the scale into Turkish, involving translation, expert panel evaluation, back-translation, and pilot testing. The Vaccine Acceptance Instrument and a sociodemographic data form were used for data collection. The reliability of the scale was tested by test–retest analysis, and its internal reliability was examined by Cronbach’s alpha test. The factor structure was examined using Exploratory Factor Analysis (EFA). Confirmatory Factor Analysis (CFA) was employed to assess the scale’s fit. Overall, 229 participants were included in the study. In test–retest reliability analysis, the intraclass correlation coefficient of the scale was 0.992 (95% CI: 0.987–0.996). The Cronbach’s alpha value of the scale was 0.824. A four-factor structure was determined. The model had an acceptable fit [χ2/df = 380.04/164 (2,317) p < 0.001, CFI = 0.91, GFI = 0.90, AGFI = 0.906, NFI = 0.90, RMSEA = 0.076]. The mean total VAI score was 112.71 ± 17.02. The low education level of the mother, being a housewife, and parents not having the COVID-19 vaccine were statistically significantly associated with a low scale score and low vaccine acceptance (p < 0.05). The Turkish adaptation of the VAI demonstrated satisfactory levels of validity and reliability following rigorous testing.
Full article
Open AccessArticle
Local Immune Activation and Age Impact on Humoral Immunity in Mice, with a Focus on IgG Sialylation
by
Priti Gupta, Tibor Sághy, Miriam Bollmann, Tao Jin, Claes Ohlsson, Hans Carlsten, Carmen Corciulo and Cecilia Engdahl
Vaccines 2024, 12(5), 479; https://doi.org/10.3390/vaccines12050479 (registering DOI) - 29 Apr 2024
Abstract
Age alters the host’s susceptibility to immune induction. Humoral immunity with circulating antibodies, particularly immunoglobulin G (IgG), plays an essential role in immune response. IgG glycosylation in the fragment crystallizable (Fc) region, including sialylation, is important in regulating the effector function by interacting
[...] Read more.
Age alters the host’s susceptibility to immune induction. Humoral immunity with circulating antibodies, particularly immunoglobulin G (IgG), plays an essential role in immune response. IgG glycosylation in the fragment crystallizable (Fc) region, including sialylation, is important in regulating the effector function by interacting with Fc gamma receptors (FcγRs). Glycosylation is fundamentally changed with age and inflammatory responses. We aimed to explore the regulation of humoral immunity by comparing responses to antigen-induced immune challenges in young and adult mice using a local antigen-induced arthritis mouse model. This study examines the differences in immune response between healthy and immune-challenged states across these groups. Our initial assessment of the arthritis model indicated that adult mice presented more severe knee swelling than their younger counterparts. In contrast, we found that neither histological assessment, bone mineral density, nor the number of osteoclasts differs. Our data revealed an age-associated but not immune challenge increase in total IgG; the only subtype affected by immune challenge was IgG1 and partially IgG3. Interestingly, the sialylation of IgG2b and IgG3 is affected by age and immune challenges but not stimulated further by immune challenges in adult mice. This suggests a shift in IgG towards a pro-inflammatory and potentially pathogenic state with age and inflammation.
Full article
(This article belongs to the Special Issue Humoral and Cellular Response after Vaccination)
Open AccessReview
Harnessing T-Cells for Enhanced Vaccine Development against Viral Infections
by
Zhen Zhuang, Jianfen Zhuo, Yaochang Yuan, Zhao Chen, Shengnan Zhang, Airu Zhu, Jingxian Zhao and Jincun Zhao
Vaccines 2024, 12(5), 478; https://doi.org/10.3390/vaccines12050478 (registering DOI) - 29 Apr 2024
Abstract
Despite significant strides in vaccine research and the availability of vaccines for many infectious diseases, the threat posed by both known and emerging infectious diseases persists. Moreover, breakthrough infections following vaccination remain a concern. Therefore, the development of novel vaccines is imperative. These
[...] Read more.
Despite significant strides in vaccine research and the availability of vaccines for many infectious diseases, the threat posed by both known and emerging infectious diseases persists. Moreover, breakthrough infections following vaccination remain a concern. Therefore, the development of novel vaccines is imperative. These vaccines must exhibit robust protective efficacy, broad-spectrum coverage, and long-lasting immunity. One promising avenue in vaccine development lies in leveraging T-cells, which play a crucial role in adaptive immunity and regulate immune responses during viral infections. T-cell recognition can target highly variable or conserved viral proteins, and memory T-cells offer the potential for durable immunity. Consequently, T-cell-based vaccines hold promise for advancing vaccine development efforts. This review delves into the latest research advancements in T-cell-based vaccines across various platforms and discusses the associated challenges.
Full article
(This article belongs to the Special Issue Review Special Issue Series: T-cell Based Vaccine Development against Pathogen Infections)
Open AccessReview
Promising Cytokine Adjuvants for Enhancing Tuberculosis Vaccine Immunity
by
Xuezhi Cao, Yang-Xin Fu and Hua Peng
Vaccines 2024, 12(5), 477; https://doi.org/10.3390/vaccines12050477 (registering DOI) - 29 Apr 2024
Abstract
Tuberculosis, caused by Mycobacterium tuberculosis (M. tuberculosis), remains a formidable global health challenge, affecting a substantial portion of the world’s population. The current tuberculosis vaccine, bacille Calmette–Guérin (BCG), offers limited protection against pulmonary tuberculosis in adults, underscoring the critical need for
[...] Read more.
Tuberculosis, caused by Mycobacterium tuberculosis (M. tuberculosis), remains a formidable global health challenge, affecting a substantial portion of the world’s population. The current tuberculosis vaccine, bacille Calmette–Guérin (BCG), offers limited protection against pulmonary tuberculosis in adults, underscoring the critical need for innovative vaccination strategies. Cytokines are pivotal in modulating immune responses and have been explored as potential adjuvants to enhance vaccine efficacy. The strategic inclusion of cytokines as adjuvants in tuberculosis vaccines holds significant promise for augmenting vaccine-induced immune responses and strengthening protection against M. tuberculosis. This review delves into promising cytokines, such as Type I interferons (IFNs), Type II IFN, interleukins such as IL-2, IL-7, IL-15, IL-12, and IL-21, alongside the use of a granulocyte–macrophage colony-stimulating factor (GM-CSF) as an adjuvant, which has shown effectiveness in boosting immune responses and enhancing vaccine efficacy in tuberculosis models.
Full article
(This article belongs to the Special Issue Review Special Issue Series: T-cell Based Vaccine Development against Pathogen Infections)
Open AccessArticle
Evaluation of Swine Protection with Three Commercial Foot-and-Mouth Disease Vaccines against Heterologous Challenge with Type A ASIA/G-VII Lineage Viruses
by
Seon Woo Kim, Seung Heon Lee, Ha-Hyun Kim, Sung-Ho Shin, Sang-Hyun Park, Jong-Hyeon Park, Jaejo Kim and Choi-Kyu Park
Vaccines 2024, 12(5), 476; https://doi.org/10.3390/vaccines12050476 (registering DOI) - 29 Apr 2024
Abstract
Outbreaks caused by foot-and-mouth disease (FMD) A/ASIA/G-VII lineage viruses have often occurred in Middle Eastern and Southeast Asian countries since 2015. Because A/ASIA/G-VII lineage viruses are reported to have distinct antigenic relatedness with available commercial FMD vaccine strains, it is necessary to investigate
[...] Read more.
Outbreaks caused by foot-and-mouth disease (FMD) A/ASIA/G-VII lineage viruses have often occurred in Middle Eastern and Southeast Asian countries since 2015. Because A/ASIA/G-VII lineage viruses are reported to have distinct antigenic relatedness with available commercial FMD vaccine strains, it is necessary to investigate whether inoculation with vaccines used in Korea could confer cross-protection against A/ASIA/G-VII lineage viruses. In the present study, we conducted two vaccination challenge trials to evaluate the efficacy of three commercial FMD vaccines (O/Manisa + O/3039 + A/Iraq, O/Campos + A/Cruzeiro + A/2001, and O/Primorsky + A/Zabaikalsky) against heterologous challenge with ASIA/G-VII lineage viruses (A/TUR/13/2017 or A/BHU/3/2017 strains) in pigs. In each trial, clinical signs, viremia, and salivary shedding of virus were measured for 7 days after challenge. In summary, the O/Campos + A/Cruzeiro + A/2001 vaccine provided full protection against two A/ASIA/G-VII lineage viruses in vaccinated pigs, where significant protection was observed. Although unprotected animals were observed in groups vaccinated with O/Manisa + O/3039 + A/Iraq or O/Primorsky + A/Zabaikalsky vaccines, the clinical scores and viral RNA levels in the sera and oral swabs of vaccinated animals were significantly lower than those of unvaccinated controls.
Full article
(This article belongs to the Section Veterinary Vaccines)
Open AccessArticle
Anti-SARS-CoV-2 Antibodies Level and COVID-19 Vaccine Boosters among Healthcare Workers with the Highest SARS-CoV-2 Infection Risk—Follow Up Study
by
Dagny Lorent, Rafał Nowak, Magdalena Figlerowicz, Luiza Handschuh and Paweł Zmora
Vaccines 2024, 12(5), 475; https://doi.org/10.3390/vaccines12050475 (registering DOI) - 29 Apr 2024
Abstract
During the COVID-19 pandemic, several vaccines were developed to limit the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). However, due to SARS-CoV-2 mutations and uneven vaccination coverage among populations, a series of COVID-19 waves have been caused by different variants of
[...] Read more.
During the COVID-19 pandemic, several vaccines were developed to limit the spread of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). However, due to SARS-CoV-2 mutations and uneven vaccination coverage among populations, a series of COVID-19 waves have been caused by different variants of concern (VOCs). Despite the updated vaccine formulations for the new VOC, the benefits of additional COVID-19 vaccine doses have raised many doubts, even among high-risk groups such as healthcare workers (HCWs). We examined the factors underlying hesitancy to receive COVID-19 booster vaccine doses and analysed the anti-SARS-CoV-2 IgG antibody response after booster vaccination among HCWs. Our study found that 42% of the HCWs were hesitant about the second booster dose, while 7% reported no intent to get vaccinated with any additional doses. As reasons for not vaccinating, participants most frequently highlighted lack of time, negative experiences with previous vaccinations, and immunity conferred by past infections. In addition, we found the lowest post-vaccination antibody titres among HCWs who did not receive any vaccine booster dose and the highest among HCWs vaccinated with two booster doses.
Full article
(This article belongs to the Special Issue Knowledge, Attitudes, Beliefs and Hesitancy towards COVID-19 Vaccination and Booster Doses)
Open AccessArticle
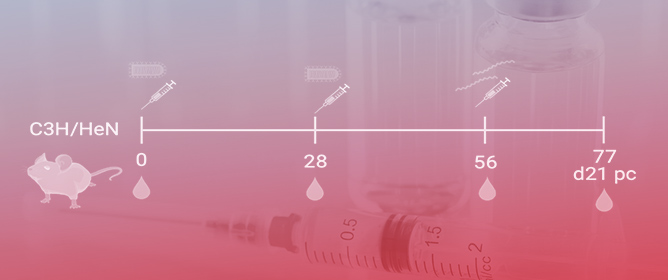
A Clinical Analysis of Anti-Programmed Death-Ligand 1 (PD-L1) Immune Checkpoint Inhibitor Treatments Combined with Chemotherapy in Untreated Extensive-Stage Small-Cell Lung Cancer
by
Ping-Chih Hsu, Bing-Chen Wu, Chin-Chou Wang, Li-Chung Chiu, Chiung-Hsin Chang, Ping-Chi Liu, Chiao-En Wu, Scott Chih-Hsi Kuo, Jia-Shiuan Ju, Allen Chung-Cheng Huang, Yu-Ching Lin, Cheng-Ta Yang and How-Wen Ko
Vaccines 2024, 12(5), 474; https://doi.org/10.3390/vaccines12050474 (registering DOI) - 29 Apr 2024
Abstract
Real-world clinical experience of using anti-programmed death-ligand 1 (PD-L1) immune checkpoint inhibitors (ICIs) combined with chemotherapy in the first-line treatment of extensive-stage small-cell lung cancer (SCLC) patients has rarely been reported. In this study, we aimed to perform a retrospective multicenter clinical analysis
[...] Read more.
Real-world clinical experience of using anti-programmed death-ligand 1 (PD-L1) immune checkpoint inhibitors (ICIs) combined with chemotherapy in the first-line treatment of extensive-stage small-cell lung cancer (SCLC) patients has rarely been reported. In this study, we aimed to perform a retrospective multicenter clinical analysis of extensive-stage SCLC patients receiving first-line therapy with anti-PD-L1 ICIs combined with chemotherapy. Between November 2018 and March 2022, 72 extensive-stage SCLC patients receiving first-line atezolizumab or durvalumab in combination with chemotherapy, according to the cancer center databases of Linkou, Chiayi, and Kaohsiung Chang Gung Memorial Hospitals, were retrospectively included in the analysis. Twenty-one patients (29.2%) received atezolizumab and fifty-one (70.8%) received durvalumab. Objective response (OR) and disease control (DC) rates of 59.7% and 73.6%, respectively, were observed with first-line ICI plus chemotherapy. The median progression-free survival (PFS) was 6.63 months (95% confidence interval (CI), 5.25–8.02), and the median overall survival (OS) was 16.07 months (95% CI, 15.12–17.0) in all study patients. A high neutrophil-to-lymphocyte ratio (NLR; >4) and a high serum lactate dehydrogenase (LDH) concentration (>260 UL) were identified as independent unfavorable factors associated with shorter OS in the multivariate analysis. Regarding safety, neutropenia was the most common grade 3 treatment-related adverse event (AE), but no treatment-related deaths occurred in the study patients. First-line anti-PD-L1 ICIs combined with chemotherapy are effective and safe for male extensive-stage SCLC patients. Further therapeutic strategies may need to be developed for patients with unfavorable outcomes (e.g., baseline high NLR and serum LDH level).
Full article
(This article belongs to the Special Issue Immune Checkpoint Inhibitor-Based Immunotherapy in Cancer: Predictive Biomarkers and Mechanisms of Resistance)
►▼
Show Figures
Figure 1
Open AccessArticle
Assessing the Impact of the COVID-19 Pandemic on Pregnant Women’s Attitudes towards Childhood Vaccinations: A Cross-Sectional Study
by
Paola Arcaro, Lorenza Nachira, Fabio Pattavina, Enrica Campo, Rossella Mancini, Domenico Pascucci, Gianfranco Damiani, Brigida Carducci, Antonietta Spadea, Antonio Lanzone, Stefania Bruno and Patrizia Laurenti
Vaccines 2024, 12(5), 473; https://doi.org/10.3390/vaccines12050473 (registering DOI) - 29 Apr 2024
Abstract
The COVID-19 pandemic has globally disrupted immunisation practices, impacting vulnerable populations such as pregnant women (PW), who harbour concerns about future children’s immunisations. This study aimed to assess the pandemic’s impact on PW’s attitudes towards childhood vaccinations. During three consecutive flu seasons from
[...] Read more.
The COVID-19 pandemic has globally disrupted immunisation practices, impacting vulnerable populations such as pregnant women (PW), who harbour concerns about future children’s immunisations. This study aimed to assess the pandemic’s impact on PW’s attitudes towards childhood vaccinations. During three consecutive flu seasons from October 2019 to January 2022, a cross-sectional study was conducted in a large Italian teaching hospital using a questionnaire. The chi-square test was performed to compare each season. Across the 2019–2020 to 2021–2022 seasons, course attendance by PW surged from 105 to 340. Significant shifts in vaccination intentions were noted, including a 7.5% decrease in measles vaccination intent (p = 0.02) and a 10% decrease in that of pertussis (p = 0.004) from 2019–2020 to 2020–2021. While perceived contagion risk decreased, disease severity perceptions increased, with few significant differences. A statistically significant reduction was noted in the proportion of participants suspecting economic motives behind NHS workers’ promotion of childhood vaccinations. Furthermore, the pandemic period saw an increase in the perceived utility of non-institutional websites and the advice of physicians outside the NHS. These findings will help develop evidence-based, tailored interventions and communication strategies to address vaccine hesitancy and ensure optimal vaccination coverage among children born during and after the pandemic.
Full article
(This article belongs to the Section Human Vaccines and Public Health)
Open AccessArticle
Construction and Mechanism Exploration of Highly Efficient System for Bacterial Ghosts Preparation Based on Engineered Phage ID52 Lysis Protein E
by
Yi Ma, Sijia Wang, Bin Hong, Lan Feng and Jufang Wang
Vaccines 2024, 12(5), 472; https://doi.org/10.3390/vaccines12050472 (registering DOI) - 28 Apr 2024
Abstract
Bacterial ghosts (BGs) are hollow bacterial cell envelopes with intact cellular structures, presenting as promising candidates for various biotechnological and biomedical applications. However, the yield and productivity of BGs have encountered limitations, hindering their large-scale preparation and multi-faceted applications of BGs. Further optimization
[...] Read more.
Bacterial ghosts (BGs) are hollow bacterial cell envelopes with intact cellular structures, presenting as promising candidates for various biotechnological and biomedical applications. However, the yield and productivity of BGs have encountered limitations, hindering their large-scale preparation and multi-faceted applications of BGs. Further optimization of BGs is needed for the commercial application of BG technology. In this study, we screened out the most effective lysis protein ID52-E-W4A among 13 mutants based on phage ID52 lysis protein E and optimized the liquid culture medium for preparing Escherichia coli Nissle 1917 (EcN). The results revealed a significantly higher lysis rate of ID52-E-W4A compared to that of ID52-E in the 2xYT medium. Furthermore, EcN BGs were cultivated in a fermenter, achieving an initial OD600 as high as 6.0 after optimization, indicating enhanced BG production. Moreover, the yield of ID52-E-W4A-induced BGs reached 67.0%, contrasting with only a 3.1% yield from φX174-E-induced BGs. The extended applicability of the lysis protein ID52-E-W4A was demonstrated through the preparation of Salmonella pullorum ghosts and Salmonella choleraesuis ghosts. Knocking out the molecular chaperone gene slyD and dnaJ revealed that ID52-mediated BGs could still undergo lysis. Conversely, overexpression of integral membrane enzyme gene mraY resulted in the loss of lysis activity for ID52-E, suggesting that the lysis protein ID52-E may no longer rely on SlyD or DnaJ to function, with MraY potentially being the target of ID52-E. This study introduces a novel approach utilizing ID52-E-W4A for recombinant expression, accelerating the BG formation and thereby enhancing BG yield and productivity.
Full article
(This article belongs to the Section Vaccines against Infectious Diseases)
►▼
Show Figures
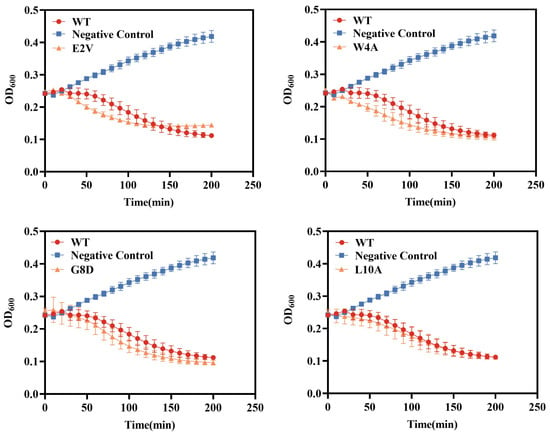
Figure 1
Open AccessArticle
High Concentration of Anti-SARS-CoV-2 Antibodies 2 Years after COVID-19 Vaccination Stems Not Only from Boosters but Also from Widespread, Often Unrecognized, Contact with the Virus
by
Jakub Swadźba, Andrzej Panek, Paweł Wąsowicz, Tomasz Anyszek and Emilia Martin
Vaccines 2024, 12(5), 471; https://doi.org/10.3390/vaccines12050471 (registering DOI) - 28 Apr 2024
Abstract
This study follows 99 subjects vaccinated with Pfizer/BioNTech COVID-19 vaccines over two years, with particular focus on the last year of observation (between days 360 and 720). The response to the vaccination was assessed with Diasorin’s SARS-CoV-2 TrimericSpike IgG. Screening for SARS-CoV-2 infection
[...] Read more.
This study follows 99 subjects vaccinated with Pfizer/BioNTech COVID-19 vaccines over two years, with particular focus on the last year of observation (between days 360 and 720). The response to the vaccination was assessed with Diasorin’s SARS-CoV-2 TrimericSpike IgG. Screening for SARS-CoV-2 infection was performed with Abbott’s SARS-CoV-2 Nucleocapsid IgG immunoassay. Data from questionnaires were also analyzed. Two years after the first vaccine dose administration, 100% of the subjects were positive for anti-spike SARS-CoV-2 IgG and the median antibody level was still high (3600 BAU/mL), dropping insignificantly over the last year. Simultaneously, a substantial increase in seropositivity in anti-nucleocapsid SARS-CoV-2 IgG was noted, reaching 33%. There was no statistically significant agreement between anti-N seropositivity and reported COVID-19. Higher anti-spike concentrations and lower COVID-19 incidence was seen in the older vaccinees. It was noted that only subjects boosted between days 360 and 720 showed an increase in anti-spike IgG concentrations. The higher antibody concentrations (median 7440 BAU/mL) on day 360 were noted in participants not infected over the following year. Vaccination, including booster administrations, and natural, even unrecognized, contact with SARS-CoV-2 entwined two years after the primary vaccination, leading to high anti-spike antibody concentrations.
Full article
(This article belongs to the Special Issue 2nd Edition of Antibody Response to Infection and Vaccination)
Open AccessArticle
Attitude to Co-Administration of Influenza and COVID-19 Vaccines among Pregnant Women Exploring the Health Action Process Approach Model
by
Alessandra Fallucca, Palmira Immordino, Patrizia Ferro, Luca Mazzeo, Sefora Petta, Antonio Maiorana, Marianna Maranto, Alessandra Casuccio and Vincenzo Restivo
Vaccines 2024, 12(5), 470; https://doi.org/10.3390/vaccines12050470 (registering DOI) - 28 Apr 2024
Abstract
Respiratory tract diseases caused by influenza virus and SARS-CoV-2 can represent a serious threat to the health of pregnant women. Immunological remodulation for fetus tolerance and physiological changes in the gestational chamber expose both mother and child to fearful complications and a high
[...] Read more.
Respiratory tract diseases caused by influenza virus and SARS-CoV-2 can represent a serious threat to the health of pregnant women. Immunological remodulation for fetus tolerance and physiological changes in the gestational chamber expose both mother and child to fearful complications and a high risk of hospitalization. Vaccines to protect pregnant women from influenza and COVID-19 are strongly recommended and vaccine co-administration could be advantageous to increase coverage of both vaccines. The attitude to accept both vaccines is affected by several factors: social, cultural, and cognitive-behavioral. In Palermo, Italy, during the 2021–2022 influenza season, a cross-sectional study was conducted to evaluate pregnant women’s intention to adhere to co-administration of influenza and COVID-19 vaccines. The determinants of vaccination attitude were investigated through the administration of a questionnaire and the Health Action Process Approach theory was adopted to explore the cognitive behavioral aspects. Overall, 120 pregnant women were enrolled; mean age 32 years, 98.2% (n = 118) of Italian nationality and 25.2% (n = 30) with obstetric or pathological conditions of pregnancy at risk. Factors significantly associated with the attitude to co-administration of influenza and COVID-19 vaccines among pregnant women were: high level of education (OR = 13.96; p < 0.001), positive outcome expectations (OR = 2.84; p < 0.001), and self-efficacy (OR = 3.1; p < 0.001). Effective strategies to promote the co-administration of the influenza vaccine and the COVID-19 vaccine should be based on the communication of the benefits and positive outcomes of vaccine co-administration and on the adequate information of pregnant women.
Full article
(This article belongs to the Special Issue Vaccination Strategies for COVID-19 II)
Open AccessArticle
Identification and Quantification of a Pneumococcal Cell Wall Polysaccharide by Antibody-Enhanced Chromatography Assay
by
James Z. Deng, Zhifeng Chen, James Small, Yue Yuan, Kara Cox, Aimin Tang, Jeanette Roman, Liming Guan, Katrina Feller, Frances Ansbro and Kalpit Vora
Vaccines 2024, 12(5), 469; https://doi.org/10.3390/vaccines12050469 (registering DOI) - 28 Apr 2024
Abstract
►▼
Show Figures
Multivalent pneumococcal vaccines have been developed successfully to combat invasive pneumococcal diseases (IPD) and reduce the associated healthcare burden. These vaccines employ pneumococcal capsular polysaccharides (PnPs), either conjugated or unconjugated, as antigens to provide serotype-specific protection. Pneumococcal capsular polysaccharides used for vaccine often
[...] Read more.
Multivalent pneumococcal vaccines have been developed successfully to combat invasive pneumococcal diseases (IPD) and reduce the associated healthcare burden. These vaccines employ pneumococcal capsular polysaccharides (PnPs), either conjugated or unconjugated, as antigens to provide serotype-specific protection. Pneumococcal capsular polysaccharides used for vaccine often contain residual levels of cell wall polysaccharides (C-Ps), which can generate a non-serotype specific immune response and complicate the desired serotype-specific immunity. Therefore, the C-P level in a pneumococcal vaccine needs to be controlled in the vaccine process and the anti C-P responses need to be dialed out in clinical assays. Currently, two types of cell-wall polysaccharide structures have been identified: a mono-phosphocholine substituted cell-wall polysaccharide C-Ps1 and a di-phosphocholine substituted C-Ps2 structure. In our effort to develop a next-generation novel pneumococcal conjugate vaccine (PCV), we have generated a monoclonal antibody (mAb) specific to cell-wall polysaccharide C-Ps2 structure. An antibody-enhanced HPLC assay (AE-HPLC) has been established for serotype-specific quantification of pneumococcal polysaccharides in our lab. With the new anti C-Ps2 mAb, we herein extend the AE-HPLC assay to the quantification and identification of C-Ps2 species in pneumococcal polysaccharides used for vaccines.
Full article
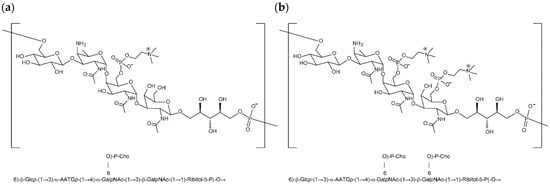
Figure 1
Open AccessSystematic Review
A Comparison of the Immunogenicity and Safety of an Additional Heterologous versus Homologous COVID-19 Vaccination among Non-Seroconverted Immunocompromised Patients after a Two-Dose Primary Series of mRNA Vaccination: A Systematic Review and Meta-Analysis
by
Chatchaya Nangsue, Karan Srisurapanont and Tavitiya Sudjaritruk
Vaccines 2024, 12(5), 468; https://doi.org/10.3390/vaccines12050468 (registering DOI) - 28 Apr 2024
Abstract
This systematic review and meta-analysis aimed to compare the immunogenicity and safety of an additional heterologous (viral vector) versus homologous (mRNA) COVID-19 vaccine dose among non-seroconverted immunocompromised patients after a two-dose primary series of mRNA vaccine. We searched studies published up to 21
[...] Read more.
This systematic review and meta-analysis aimed to compare the immunogenicity and safety of an additional heterologous (viral vector) versus homologous (mRNA) COVID-19 vaccine dose among non-seroconverted immunocompromised patients after a two-dose primary series of mRNA vaccine. We searched studies published up to 21 June 2023 in PubMed, Scopus, and Embase. The meta-analysis was conducted to compare the seropositivity rates based on anti-SARS-CoV-2 spike protein IgG (anti-S IgG) and SARS-CoV-2-specific T-cell immune response rates, assessed by interferon-γ release assay at 4 weeks, and the incidences of serious adverse events (SAEs) within 28 days between the two vaccine regimens. In four included randomized controlled trials (RCTs), there were no statistically significant differences in the seropositive rate of anti-S IgG (risk ratio [RR]: 0.79, 95% CI: 0.48–1.29) and the concentration of SARS-CoV-2 interferon-γ (RR: 1.19, 95% CI: 0.96–1.48) between heterologous and homologous regimens. The heterologous regimen exhibited a significantly lower incidence of injection pain (RR: 0.55, 95% CI: 0.45–0.69), but a higher incidence of headache (RR: 1.44, 95% CI: 1.02–2.02) compared with the homologous regimen. No vaccine-related SAEs were reported within 28 days following vaccination. An additional heterologous or homologous COVID-19 vaccine dose was well tolerated and demonstrated a comparable vaccine immunogenicity among non-seroconverted immunocompromised patients who were initially vaccinated with a two-dose COVID-19 mRNA vaccine. This finding supports the recommendations of an extended primary series of COVID-19 vaccination in immunocompromised persons.
Full article
(This article belongs to the Section COVID-19 Vaccines and Vaccination)
►▼
Show Figures
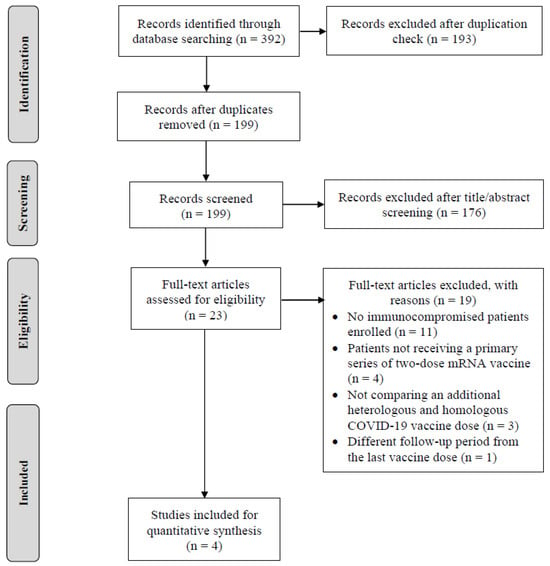
Figure 1
Open AccessReview
The D Gene in CDR H3 Determines a Public Class of Human Antibodies to SARS-CoV-2
by
Meng Yuan and Ian A. Wilson
Vaccines 2024, 12(5), 467; https://doi.org/10.3390/vaccines12050467 (registering DOI) - 27 Apr 2024
Abstract
Public antibody responses have been found against many infectious agents. Structural convergence of public antibodies is usually determined by immunoglobulin V genes. Recently, a human antibody public class against SARS-CoV-2 was reported, where the D gene (IGHD3-22) encodes a common YYDxxG motif in
[...] Read more.
Public antibody responses have been found against many infectious agents. Structural convergence of public antibodies is usually determined by immunoglobulin V genes. Recently, a human antibody public class against SARS-CoV-2 was reported, where the D gene (IGHD3-22) encodes a common YYDxxG motif in heavy-chain complementarity-determining region 3 (CDR H3), which determines specificity for the receptor-binding domain (RBD). In this review, we discuss the isolation, structural characterization, and genetic analyses of this class of antibodies, which have been isolated from various cohorts of COVID-19 convalescents and vaccinees. All eleven YYDxxG antibodies with available structures target the SARS-CoV-2 RBD in a similar binding mode, where the CDR H3 dominates the interaction with antigen. The antibodies target a conserved site on the RBD that does not overlap with the receptor-binding site, but their particular angle of approach results in direct steric hindrance to receptor binding, which enables both neutralization potency and breadth. We also review the properties of CDR H3-dominant antibodies that target other human viruses. Overall, unlike most public antibodies, which are identified by their V gene usage, this newly discovered public class of YYDxxG antibodies is dominated by a D-gene-encoded motif and uncovers further opportunities for germline-targeting vaccine design.
Full article
(This article belongs to the Special Issue Infectious Diseases: Antibodies and Vaccines)
►▼
Show Figures
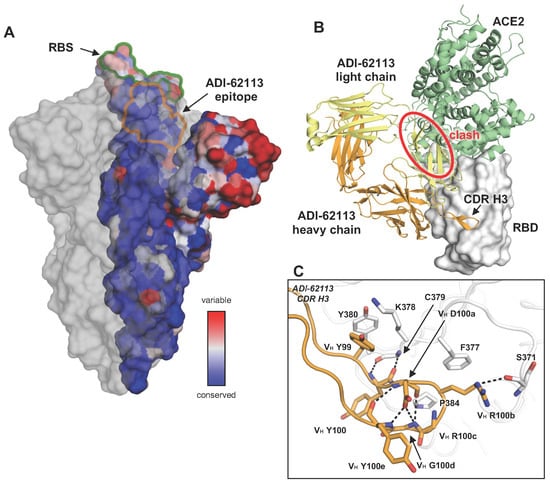
Figure 1
Open AccessArticle
Severity of Omicron Subvariants and Vaccine Impact in Catalonia, Spain
by
Víctor López de Rioja, Luca Basile, Aida Perramon-Malavez, Érica Martínez-Solanas, Daniel López, Sergio Medina Maestro, Ermengol Coma, Francesc Fina, Clara Prats, Jacobo Mendioroz Peña and Enric Alvarez-Lacalle
Vaccines 2024, 12(5), 466; https://doi.org/10.3390/vaccines12050466 (registering DOI) - 27 Apr 2024
Abstract
In the current COVID-19 landscape dominated by Omicron subvariants, understanding the timing and efficacy of vaccination against emergent lineages is crucial for planning future vaccination campaigns, yet detailed studies stratified by subvariant, vaccination timing, and age groups are scarce. This retrospective study analyzed
[...] Read more.
In the current COVID-19 landscape dominated by Omicron subvariants, understanding the timing and efficacy of vaccination against emergent lineages is crucial for planning future vaccination campaigns, yet detailed studies stratified by subvariant, vaccination timing, and age groups are scarce. This retrospective study analyzed COVID-19 cases from December 2021 to January 2023 in Catalonia, Spain, focusing on vulnerable populations affected by variants BA.1, BA.2, BA.5, and BQ.1 and including two national booster campaigns. Our database includes detailed information such as dates of diagnosis, hospitalization and death, last vaccination, and cause of death, among others. We evaluated the impact of vaccination on disease severity by age, variant, and vaccination status, finding that recent vaccination significantly mitigated severity across all Omicron subvariants, although efficacy waned six months post-vaccination, except for BQ.1, which showed more stable levels. Unvaccinated individuals had higher hospitalization and mortality rates. Our results highlight the importance of periodic vaccination to reduce severe outcomes, which are influenced by variant and vaccination timing. Although the seasonality of COVID-19 is uncertain, our analysis suggests the potential benefit of annual vaccination in populations >60 years old, probably in early fall, if COVID-19 eventually exhibits a major peak similar to other respiratory viruses.
Full article
(This article belongs to the Special Issue COVID-19 and Vaccination Strategies in Global Health)
►▼
Show Figures
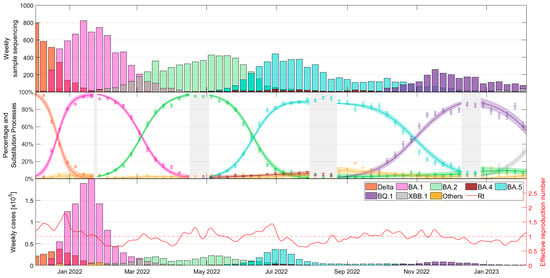
Figure 1
Open AccessSystematic Review
New Onset and Exacerbation of Autoimmune Bullous Dermatosis Following COVID-19 Vaccination: A Systematic Review
by
Po-Chien Wu, I-Hsin Huang, Ching-Ya Wang and Ching-Chi Chi
Vaccines 2024, 12(5), 465; https://doi.org/10.3390/vaccines12050465 (registering DOI) - 26 Apr 2024
Abstract
Background: Cases of autoimmune bullous dermatosis (AIBD) have been reported following COVID-19 vaccination. Objective: We aimed to provide an overview of clinical characteristics, treatments, and outcomes of AIBDs following COVID-19 vaccination. Methods: We conducted a systematic review and searched the Embase, Cochrane Library,
[...] Read more.
Background: Cases of autoimmune bullous dermatosis (AIBD) have been reported following COVID-19 vaccination. Objective: We aimed to provide an overview of clinical characteristics, treatments, and outcomes of AIBDs following COVID-19 vaccination. Methods: We conducted a systematic review and searched the Embase, Cochrane Library, and Medline databases from their inception to 27 March 2024. We included all studies reporting ≥ 1 patient who developed new-onset AIBD or experienced flare of AIBD following at least one dose of any COVID-19 vaccine. Results: We included 98 studies with 229 patients in the new-onset group and 216 in the flare group. Among the new-onset cases, bullous pemphigoid (BP) was the most frequently reported subtype. Notably, mRNA vaccines were commonly associated with the development of AIBD. Regarding the flare group, pemphigus was the most frequently reported subtype, with the mRNA vaccines being the predominant vaccine type. The onset of AIBD ranged from 1 to 123 days post-vaccination, with most patients displaying favorable outcomes and showing improvement or resolution from 1 week to 8 months after treatment initiation. Conclusions: Both new-onset AIBD and exacerbation of pre-existing AIBD may occur following COVID-19 vaccination. Healthcare practitioners should be alert, and post-vaccination monitoring may be essential.
Full article
(This article belongs to the Section COVID-19 Vaccines and Vaccination)
Open AccessArticle
Longitudinal Analysis of Humoral and Cellular Immune Response up to 6 Months after SARS-CoV-2 BA.5/BF.7/XBB Breakthrough Infection and BA.5/BF.7-XBB Reinfection
by
Xun Wang, Meng Zhang, Kaifeng Wei, Chen Li, Jinghui Yang, Shujun Jiang, Chaoyue Zhao, Xiaoyu Zhao, Rui Qiao, Yuchen Cui, Yanjia Chen, Jiayan Li, Guonan Cai, Changyi Liu, Jizhen Yu, Wenhong Zhang, Faren Xie, Pengfei Wang and Yanliang Zhang
Vaccines 2024, 12(5), 464; https://doi.org/10.3390/vaccines12050464 - 26 Apr 2024
Abstract
The rapid mutation of SARS-CoV-2 has led to multiple rounds of large-scale breakthrough infection and reinfection worldwide. However, the dynamic changes of humoral and cellular immunity responses to several subvariants after infection remain unclear. In our study, a 6-month longitudinal immune response evaluation
[...] Read more.
The rapid mutation of SARS-CoV-2 has led to multiple rounds of large-scale breakthrough infection and reinfection worldwide. However, the dynamic changes of humoral and cellular immunity responses to several subvariants after infection remain unclear. In our study, a 6-month longitudinal immune response evaluation was conducted on 118 sera and 50 PBMC samples from 49 healthy individuals who experienced BA.5/BF.7/XBB breakthrough infection or BA.5/BF.7-XBB reinfection. By studying antibody response, memory B cell, and IFN-γ secreting CD4+/CD8+ T cell response to several SARS-CoV-2 variants, we observed that each component of immune response exhibited distinct kinetics. Either BA.5/BF.7/XBB breakthrough infection or BA.5/BF.7-XBB reinfection induces relatively high level of binding and neutralizing antibody titers against Omicron subvariants at an early time point, which rapidly decreases over time. Most of the individuals at 6 months post-breakthrough infection completely lost their neutralizing activities against BQ.1.1, CH.1.1, BA.2.86, JN.1 and XBB subvariants. Individuals with BA.5/BF.7-XBB reinfection exhibit immune imprinting shifting and recall pre-existing BA.5/BF.7 neutralization antibodies. In the BA.5 breakthrough infection group, the frequency of BA.5 and XBB.1.16-RBD specific memory B cells, resting memory B cells, and intermediate memory B cells gradually increased over time. On the other hand, the frequency of IFN-γ secreting CD4+/CD8+ T cells induced by WT/BA.5/XBB.1.16 spike trimer remains stable over time. Overall, our research indicates that individuals with breakthrough infection have rapidly declining antibody levels but have a relatively stable cellular immunity that can provide some degree of protection from future exposure to new antigens.
Full article
(This article belongs to the Special Issue The Role of B Cells and Antibodies against Infectious Diseases)
Open AccessArticle
Enhancing COVID-19 Vaccine Uptake among Tribal Communities: A Case Study on Program Implementation Experiences from Jharkhand and Chhattisgarh States, India
by
Ankita Meghani, Manjula Sharma, Tanya Singh, Sourav Ghosh Dastidar, Veena Dhawan, Natasha Kanagat, Anil Gupta, Anumegha Bhatnagar, Kapil Singh, Jessica C. Shearer and Gopal Krishna Soni
Vaccines 2024, 12(5), 463; https://doi.org/10.3390/vaccines12050463 - 26 Apr 2024
Abstract
Tribal populations in India have health care challenges marked by limited access due to geographical distance, historical isolation, cultural differences, and low social stratification, and that result in weaker health indicators compared to the general population. During the pandemic, Tribal districts consistently reported
[...] Read more.
Tribal populations in India have health care challenges marked by limited access due to geographical distance, historical isolation, cultural differences, and low social stratification, and that result in weaker health indicators compared to the general population. During the pandemic, Tribal districts consistently reported lower COVID-19 vaccination coverage than non-Tribal districts. We assessed the MOMENTUM Routine Immunization Transformation and Equity (the project) strategy, which aimed to increase access to and uptake of COVID-19 vaccines among Tribal populations in Chhattisgarh and Jharkhand using the reach, effectiveness, adoption, implementation, and maintenance framework. We designed a qualitative explanatory case study and conducted 90 focus group discussions and in-depth interviews with Tribal populations, community-based nongovernmental organizations that worked with district health authorities to implement the interventions, and other stakeholders such as government and community groups. The active involvement of community leaders, targeted counseling, community gatherings, and door-to-door visits appeared to increase vaccine awareness and assuage concerns about its safety and efficacy. Key adaptations such as conducting evening vaccine awareness activities, holding vaccine sessions at flexible times and sites, and modifying messaging for booster doses appeared to encourage vaccine uptake among Tribal populations. While we used project resources to mitigate financial and supply constraints where they arose, sustaining long-term uptake of project interventions appears dependent on continued funding and ongoing political support.
Full article
(This article belongs to the Special Issue Inequality in Immunization 2024)
Open AccessArticle
The Burden of Herpes Zoster on Hospital Admissions: A Retrospective Analysis in the Years of 2015–2021 from the Abruzzo Region, Italy
by
Piera Scampoli, Giuseppe Di Martino, Fabrizio Cedrone, Camillo Odio, Pamela Di Giovanni, Ferdinando Romano and Tommaso Staniscia
Vaccines 2024, 12(5), 462; https://doi.org/10.3390/vaccines12050462 - 26 Apr 2024
Abstract
(1) Background: Herpes zoster (HZ) is a disease caused by the reactivation of the Varicella Zoster Virus (VZV). Clinical reactivation, herpes zoster, takes place in 10–20% of subjects who contracted the primary infection, with a higher risk of developing zoster increasing proportionally with
[...] Read more.
(1) Background: Herpes zoster (HZ) is a disease caused by the reactivation of the Varicella Zoster Virus (VZV). Clinical reactivation, herpes zoster, takes place in 10–20% of subjects who contracted the primary infection, with a higher risk of developing zoster increasing proportionally with age, especially after 50 years of age. HZ is a common clinical problem, particularly among patients aged over 50 years and immunocompromised patients. Immunocompromised patients and adults could present an atypical and more severe course. In addition, they are at greater risk of complications. For this reason, it is important to understand the real burden of the disease and to identify the subjects who are at higher risk of HZ and its complications, also to direct preventive strategies at the right targets. The aim of the present study is to analyze HZ-related hospitalization trends in Abruzzo in the period of 2015–2021. (2) Methods: Data related to hospital admissions were extracted from the hospital discharge records (HDRs) of the whole region, considering all admissions during the years of 2015–2021. The trends in hospital admissions and length of stay were evaluated and analyzed. (3) Results: A total of 768 hospital discharges with a diagnosis of herpes zoster were registered in Abruzzo during the 7-year study period. During the study period, an increasing trend was observed from the year 2015 to the year 2017, ranging from 8.19 cases/100,000 to 11.5 cases/100,000 (APC (Annual percentage change) +20.8%; 95%CI −2.3; 47.6). After the year 2017, a significantly decreasing trend was observed, reaching 5.46 cases/100,000 in the year 2021 (APC −18.4%; 95%CI −31.5; −12.0). Across the entire study period, an average annual percentage change (AAPC) of −7.0% (95%CI −13.0; −1.3) was observed. (4) Conclusions: Despite the trend of a reduction in hospitalizations, this study highlights that HZ continues to have a great impact on public health. So, it is important to update recommendations for the use of the already available HZ vaccine and to implement new strategies to increase awareness of the prevention of the disease.
Full article
(This article belongs to the Special Issue Varicella and Zoster Vaccination)
►▼
Show Figures
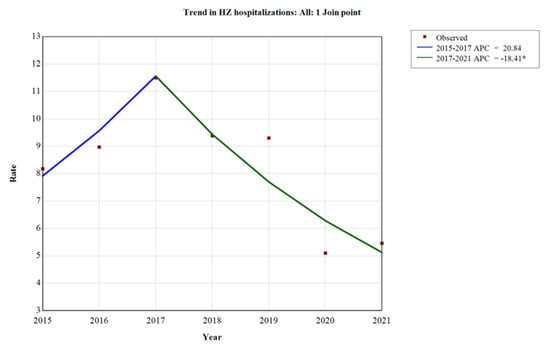
Figure 1
Open AccessArticle
The Bacterial Meningitis Epidemic in Banalia in the Democratic Republic of Congo in 2021
by
Andre Arsene Bita Fouda, Anderson Latt, Abdoulaye Sinayoko, Franck Fortune Roland Mboussou, Lorenzo Pezzoli, Katya Fernandez, Clement Lingani, Berthe Miwanda, Dorothée Bulemfu, Francis Baelongandi, Patrick Mbenga Likita, Marie-José Kikoo Bora, Marcel Sabiti, Gervais Leon Folefack Tengomo, Eugène Kabambi Kabangu, Guy Kalambayi Kabamba, Issifou Alassani, Muhamed-Kheir Taha, Ado Mpia Bwaka, Charles Shey Wiysonge and Benido Impoumaadd
Show full author list
remove
Hide full author list
Vaccines 2024, 12(5), 461; https://doi.org/10.3390/vaccines12050461 - 25 Apr 2024
Abstract
Background: The Banalia health zone in the Democratic Republic of Congo reported a meningitis epidemic in 2021 that evolved outside the epidemic season. We assessed the effects of the meningitis epidemic response. Methods: The standard case definition was used to identify cases. Care
[...] Read more.
Background: The Banalia health zone in the Democratic Republic of Congo reported a meningitis epidemic in 2021 that evolved outside the epidemic season. We assessed the effects of the meningitis epidemic response. Methods: The standard case definition was used to identify cases. Care was provided to 2651 in-patients, with 8% of them laboratory tested, and reactive vaccination was conducted. To assess the effects of reactive vaccination and treatment with ceftriaxone, a statistical analysis was performed. Results: Overall, 2662 suspected cases of meningitis with 205 deaths were reported. The highest number of cases occurred in the 30–39 years age group (927; 38.5%). Ceftriaxone contributed to preventing deaths with a case fatality rate that decreased from 70.4% before to 7.7% after ceftriaxone was introduced (p = 0.001). Neisseria meningitidis W was isolated, accounting for 47/57 (82%), of which 92% of the strains belonged to the clonal complex 11. Reactive vaccination of individuals in Banalia aged 1–19 years with a meningococcal multivalent conjugate (ACWY) vaccine (Menactra®) coverage of 104.6% resulted in an 82% decline in suspected meningitis cases (incidence rate ratio, 0.18; 95% confidence interval, 0.02–0.80; p = 0.041). Conclusion: Despite late detection (two months) and reactive vaccination four months after crossing the epidemic threshold, interventions implemented in Banalia contributed to the control of the epidemic.
Full article
(This article belongs to the Special Issue Vaccine Coverage and Safety in Immunization Programs)
►▼
Show Figures
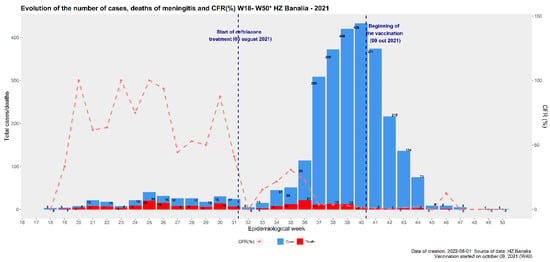
Figure 1

Journal Menu
► ▼ Journal Menu-
- Vaccines Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections & Collections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Society Collaborations
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Diseases, IJMS, Microbiology Research, Pathogens, Vaccines
Advances in Human Pathogen Control—a 21st Century Challenge 2.0
Topic Editors: Jorge H. Leitão, Nitin Amdare, Joana R FelicianoDeadline: 30 June 2024
Topic in
Cells, Diseases, Healthcare, IJMS, Vaccines
Inflammation: The Cause of all Diseases 2.0
Topic Editors: Vasso Apostolopoulos, Jack Feehan, Vivek P. ChavdaDeadline: 31 July 2024
Topic in
Biomedicines, JCM, Pathogens, Vaccines, Viruses
Discovery and Development of Monkeypox Disease Treatments
Topic Editors: Mohd Imran, Ali A. RabaanDeadline: 31 August 2024
Topic in
Brain Sciences, Clinics and Practice, COVID, Life, Vaccines, Viruses
Multifaceted Efforts from Basic Research to Clinical Practice in Controlling COVID-19 Disease
Topic Editors: Yih-Horng Shiao, Rashi OjhaDeadline: 30 September 2024

Conferences
Special Issues
Special Issue in
Vaccines
Public Psychobehavioral Responses towards Vaccination
Guest Editor: Li Ping WongDeadline: 30 April 2024
Special Issue in
Vaccines
Novel Vaccines for Infectious Pathogens
Guest Editors: Veerupaxagouda Patil, Dhruv DesaiDeadline: 20 May 2024
Special Issue in
Vaccines
COVID-19 Vaccine Acceptance and Uptake: Insights from Behavioural and Social Sciences
Guest Editor: Christian NapoliDeadline: 31 May 2024
Special Issue in
Vaccines
SARS-CoV-2 Variants, Vaccines, and Immune Responses
Guest Editor: Gianni Gori SavelliniDeadline: 15 June 2024
Topical Collections
Topical Collection in
Vaccines
COVID-19 Vaccine Hesitancy: Correlates and Interventions
Collection Editors: Manoj Sharma, Kavita Batra
Topical Collection in
Vaccines
Topic Advisory Panel Members’ Collection Series: Immunization and Vaccines for Infectious Diseases
Collection Editors: Shumaila Hanif, Ravinder Kumar
Topical Collection in
Vaccines
Research on Monoclonal Antibodies and Antibody Engineering
Collection Editor: Tatsuya Yamazaki